The Electromagnetic (EM) Spectrum
In order to understand more about UV light we must first examine the electromagnetic spectrum in more detail. Our Sun emits a wide range of solar radiation, referred to as the electromagnetic spectrum. This EM radiation includes visible light, infrared, radio, microwaves and ultraviolet light (UV). Artificial weathering attempts to simulate either th UV portion of this spectrum or the UV plus visible portion of the spectrum.
EM radiation travels as a wave, with a characteristic wavelength and frequency (λ and f respectively). These properties are related to each other by the speed of the wave, c (see equation below).
c= λ f
The speed of an EM wave in a vacuum is the speed of light, 3 x 108 ms-1. Each type of EM radiation refers to a band of specific wavelengths and frequencies, as shown below. The shorter the wavelength the higher the frequency.
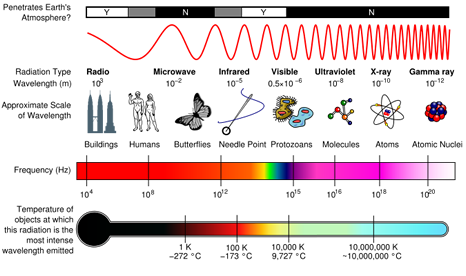
The visible and UV portion of this spectrum is expanded below to demonstrate the different types of UV bands. These are classed according to the draft standard ISO-DIS-21348.
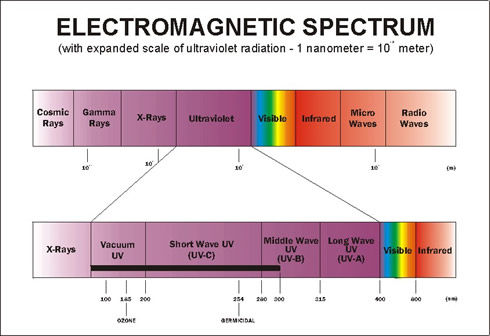
Ultraviolet light (UV) is that portion of the spectrum of invisible light below 400nm and down to 10nm. As the name indicates "beyond violet". While the primary source of UV is the sun, other sources include welder's flash, VDT's (Video display terminals), flourescent lightning, high intensity mercury vapor lamps (for night sports and high crime areas), xenon arc lamps, and other lightning. As can be seen the UV spectrum is divided into several regions. Of primary interest are the first three bands designated as A, B, and C.
A = 315 - 400 nm, B=280-315 nm, C=200-280 nm
Note: These UV bands should not be confused with the bulb type classifications used within the weathering machines as there is no correlation.
98.7% of the UV radiation emitted by the sun is blocked by the earths ozone layer and upper atosphere. The majority of radiation reaching the earths surface is UVA and a very small amount of UVB. This is demonstrated in the diagram below which shows the filtering effect of the earths atmosphere.
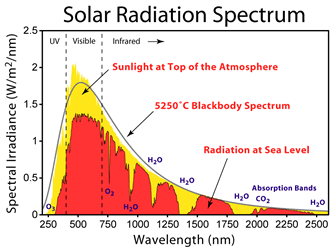
The black curve shows the emission from a perfect black body whereas the yellow region shows the normal sunlight spectrum above the earths atmosphere. The red curve gives the typical spectral irradiance at sea level. It should be noted that spectral irradiance is measured in W/m2/nm. Integration of this curve i.e. the area under the curve, provides the total irradiance.
The UVB is the component that is mainly responsible for sunburn and snowblindness wheareas the UVA portion is the element mainly responsible for degradation of polymers. The shorter the wavelength the higher the frequency and the higher the frequency the more energy the wave possess (Planck's Law). Hence shorter wavelength light tends to be the most damaging.
Ultraviolet light is considered to be the most significant with regards to non-biological material degradation, for two reasons:
1. It is of high enough wavelength to interact with molecules. The vast majority of the lower wavelength radiation, such as X-rays and γ rays are either comfortably absorbed or pass through the material. Many chemical groups found in plastic and rubber materials have absorbances within the UV band.
2. UV waves have relatively high frequencies, and consequently possess large amounts of energy, according to Planck’s Law (E=hf).
Following from the above it can be seen that the focus of most accelerated weathering machines is concentrated on the 315-400 nm range. Whilst some degradation or colour fading can also be caused by longer wavelengths the majority of damage occurs within these lower wavelengths.
